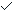4 按临床标准治愈结核的18F-FDG-PET/CT表现
目前尚没有绝对治愈并消除结核的方法。目前标准治疗期6个月是按照可接受的治疗失败率和停止药物治疗后可接受的疾病复发率来确定的[26]。在肺结核药物治疗达到痰培养阴性后,结核菌仍然可能在肺组织中持续存活多年。Malherbe等[27]报道了一项包含113例 HIV阴性的结核病患者的南非和韩国的合作研究,该研究在抗结核药物治疗之前、期间和之后不同时间点进行了18F-FDG-PET/CT扫描。在治疗结束后,该研究发现已经达到临床治愈的一部分患者可以表现有不同程度的18F-FDG摄取。在一些患者治疗前看到的高代谢病变在治疗结束后完全消退。在另外一些患者大多数病变消退,但是少数病灶可见仍然稍稍高于背景参考标准的18F-FDG摄取。而再有一些患者,虽然达到并维持了临床治愈标准,一些病变比治疗前扫描摄取18F-FDG更强烈或者出现新的高摄取病变。无论药物敏感或者耐药、持续痰培养阴性、或临床治愈,结核治疗结束时或者治疗结束1年以后,都可以看到这些动态改变。新的结核病变可能是源自于不同结核病病灶的不同治疗反应及病灶中结核亚群的变化。这些发现对解释治疗结束时18F-FDG扫描的结果提出了挑战。存活的结核菌即使在临床治愈性治疗后也常常持续存在,而且可以引发宿主反应导致18F-FDG高摄取。愈合的陈旧肺结核病通常在胸部X线片和CT上显示为纤维化及瘢痕结节,而且这些表现通常在临床治愈后持续很长时间。
有报道[28]提示有纤维化瘢痕的LTBI个体再发生活动性结核的风险增高达15倍。Jeong等[29]报道了一项研究涉及63名有放射学陈旧性愈合肺结核的患者,其中9名患者在陈旧愈合病变发现18F-FDG高摄取,其SUVmax为1.5或更高[29] (图-4)。这些代谢活跃的部位不一定代表活动性疾病,更可能反映宿主免疫反应与结核菌复制之间的平衡,但也同时代表活动性结核病发展的风险增加。


图-4。两名陈旧愈合结核受试者的胸部CT和18F-FDG-PET / CT图像, 显示高18F-FDG摄取。一例没有结核病史的76岁男性的胸部CT(左上)上观察到纤维化瘢痕和钙化结节,表明右上叶有愈合的结核灶。18F-FDG PET / CT(右上)测量病变SUVmax为4.0。TST和IGRA均为阴性。另一名71岁男性胸部CT(左下)观察到右上叶的纤维化瘢痕和结节。此患者25年前接受过肺结核治疗。18F-FDG PET / CT(右下)测量病变SUVmax为2.2。TST和IGRA均为阳性。TST: tuberculin skin test; IGRA: interferon-γ release assay。(图片来自参考文献 29)
上述证据表明,在解释治疗结束时的18F-FDGPET/CT扫描结果时,必须密切参考临床资料。18F-FDG的高摄取既可能代表持续的活动性疾病,也可能代表宿主免疫活动而且这些免疫活动会最后限制结核的发展。
5 18F-FDG-PET/CT 用于结核疗效评估
结核菌的缓慢生长而需要较长时间的治疗,这使得治疗可能由于依从性不好而失败。根据WHO指南,无耐药的单纯肺结核或淋巴结结核的标准治疗时间为6个月,复发率小于5%[30],而肺外结核的治疗时间并不统一。早期评估治疗效果是结核临床处理中关键的一环, 这对于没有细菌学诊断证据的患者以及多药耐药(multidrug resistance ,MDR) 或广泛耐药(extensive drug resistance,XDR)的结核病患者尤为重要。对细 菌学阳性的结核病患者,治疗反应观察主要通过连续细菌学检查,而对初始细菌学阴性的结核病患者的治疗反应观察通常通过临床或影像学。高达20%的肺结核患者初始细菌学可为阴性,大多数肺外结核患者也无法获得用于细菌学诊断的液体。18F-FDG-PET/CT可以在结核病X线及CT特征发生改变以前早期评估抗结核治疗反应[1,31-32]。在一项包含28名多药耐药患者研究中,Chen等[33]报道一些影像学指标可以比传统的痰微生物学更敏感地较早期区分治疗成功或不成功,其结果显示18F-FDG-PET/CT是早期预测治疗结果和长期预后的最佳方法,治疗开始2个月时18F-FDG-PET/CT预测治疗成功率的敏感性为96%,预测治疗失败的特异性为79%。CT也可以达到类似的预测率,但需要到6个月的扫描才能达到[33]。
Martinez等[34]报道了18F-FDG-PET/CT用于21例HIV阴性的结核病患者治疗评估。他们提出治疗第1个月SUVmax降低是病变改善的早期标志,而且对于无细胞学结果的病例可以验证结核病的诊断 (图-5)。如果治疗过程中的重复PET/CT的发现SUVmax较高,则应考虑缺乏依从性、耐药性或误诊的可能性。Stelzmueller等[11]报道了35例包括肺结核及肺外结核的患者,在平均治疗16.1个月后,15例患者持续存在病变18F-FDG摄取,而4例患者出现进展性病变;18F-FDG-PET和CT在初始评估和随访期间提供了相互补充的信息。Lefebvre等[35]报道,在18例淋巴结结核患者中,18F-FDG PET/CT正确识别了9例患者的治愈(治疗后PET扫描无18F-FDG摄取)和2例患者的治疗失败。应该注意的是,由于测量误差的可能性,SUVma x变异变化量小于20%~25%可能在临床上并没有意义[36]。

图-5,一名35岁女性多药耐药(MDR)结核患者抗结核治疗之前(A)和之后1个月(B)的18F-FDG PET / CT。在随访扫描中观察到肺和淋巴结病灶的减退(B)。(图片来自参考文献 34)
一些没有空洞的肺结核瘤患者可能没有临床症状而且痰培养阴性。虽然大多数肺结核瘤在抗结核治疗期间甚至治疗后变小,但也可以观察到治疗早期的一过性增大[37-38]。使用18F-FDG-PET/CT成像可能有助于这些病例的处理。如果病变显示18F-FDG摄取减少,抗结核治疗可能对结核瘤有效;而如果18F-FDG PET/CT显示病变摄取增加,则可能需要考虑改变治疗方案。
同时患有结核与艾滋病的患者可能抗结核治疗在先,然后开始抗病毒治疗。抗病毒治疗诱发的免疫反应可能导致结核病灶的炎症反应加重,这在18F-FDG-PET/CT上可能被误读为抗结核治疗反应效果欠佳[3,39]。对于这样的情况需仔细了解开始抗病毒治疗的时间,以正确解释用于评估抗结核治疗的18F-FDG-PET/CT图像。另外需要注意的是,结核病淋巴结对18F-FDG的摄取在艾滋病病毒感染者可明显高于无艾滋病的结核患者[40]。
因此,18F-FDG-PET/CT不仅是一种有用的临床工具,还有助于了解结核的病理生理学和结核感染的自然动态过程。18F-FDG-PET/CT在结核分期、肺外结核定位、识别亚临床结核患者及评估早期治疗反应方面具有重要价值。然而,18F-FDG-PET/CT成本高并且涉及辐射暴露,而且对于定性病变诊断常常缺乏特异性,因此并非所有结核患者均需要使用这个检查。尽管其作用日益增大,但18F-FDG-PET/CT在结核病诊疗中的临床应用指南尚未制定。
参考文献
1. World Health Organization: Tuberculosis.
Available at: https://www./news-room/fact-sheets/detail/tuberculosis. Accessed May 22, 2019
2. Ramirez-Lapausa M, Menendez-Saldana A, Noguerado-Asensio A. Extrapulmonary tuberculosis: an overview. Rev Esp Sanid Penit 2015; 17: 3-11
3. Ankrah AO, Glaudemans AWJM, Maes A, et al. Tuberculosis. Semin Nucl Med. 2018;48:108-130.
4. Vorster M, Sathekge MM, Bomanji J. Advances in imaging of tuberculosis: the role of ¹⁸F-FDG PET and PET/CT. Curr Opin Pulm Med. 2014;20:287-93.
5. Ankrah AO, van der Werf TS, de Vries EF, et al. PET/CT imaging of Mycobacterium tuberculosis infection. Clin Transl Imaging. 2016;4:131-144.
6. Soussan M, Brillet PY, Mekinian A, et al. Patterns of pulmonary tuberculosis on FDG-PET/CT. Eur J Radiol. 2012;81:2872-6.
7. Kim IJ, Lee JS, Kim SJ, et al: Double-phase 18F-FDG PET-CT for determination of pulmonary tuberculoma activity. Eur J Nucl Med Mol Imaging 2008; 35:808-814,
8. Goo JM, Im JG, Do KH, et al: Pulmonary tuberculoma evaluated by means of FDG PET: Findings in 10 cases. Radiology 216:117-121, 2000
9. SathekgeM, Maes A, KgomoM, et al. Impact of FDG PET on the management of TBC treatment. A pilot study. Nuklearmedizin. 2010;49:35–40.
10. Sathekge MM, Maes A, Pottel H, et al. Dual time-point FDG PET-CT for differentiating benign from malignant solitary pulmonary nodules in a TB endemic area. S Afr Med J. 2010;100:598-601.
11. Stelzmueller I, Huber H, Wunn R, et al. 18F-FDG PET/CT in the Initial Assessment and for Follow-up in Patients With Tuberculosis. Clin Nucl Med. 2016;41:e187-94.
12. Ankrah AO, Glaudemans AW, Klein HC, et al: The role of nuclear medicine in the staging and management of human immune deficiency virus infection and associated diseases. Nucl Med Mol Imaging 51:127-139, 2017
13. Salgame P, Geadas C, Collins L, et al. Latent tuberculosis infection--Revisiting and revising concepts. Tuberculosis (Edinb). 2015;95:373-84.
14. Barry CE, Boshoff HI, Dartois V, et al. The spectrum of latent tuberculosis: rethinking the biology and intervention strategies. Nature Reviews 2009, 7:845–855.
15. Lin PL, Flynn JL: Understanding latent tuberculosis: a moving target. J Immunol 2010, 185:15–22
16. Via LE, Schimel D, Weiner DM, et al: Infection dynamics and response to chemotherapy in a rabbit model of tuberculosis using [18 F]2-Fluoro- Deoxy-D-Glucose Positron Emission Tomography and Computed Tomography. Antimicrob Agents Chemother 2012, 56(8):4391
17. Gill WP, Harik NS, Whiddon MR, et al. A replication clock for Mycobacterium tuberculosis. Nat Med 2009; 15: 211-4.
18. Ford CB, Lin PL, Chase MR, et al. Use of whole genome sequencing to estimate the mutation rate of Mycobacterium tuberculosis during latent infection. Nat Genet 2011; 43: 482-6.
19. Kwan A, Seltzer M, Czernin J, et al. Characterization of hilar lymph node by 18F-fluoro-2-deoxyglucose positron emission tomography in healthy subjects. Anticancer Res 2001;21(1B):701e6.
20. Kim D-W, Kim CG. Dual-time point positron emission tomography findings of benign mediastinal lymph nodes in a tuberculosis-endemic region. Jpn J Radiol 2011;29(10):682-7.
21. Kumar A, Farhana A, Guidry L, et al. Redox homeostasis in mycobacteria: the key to tuberculosis control? Exp Rev Mol Med 2011; 13:e39.
22. Sterling TR, Villarino ME, Borisov AS, et al. Three months of rifapentine and isoniazid for latent tuberculosis infection. N Engl J Med 2011; 365:2155–2166.
23. Latent Tuberculosis Infection: A Guide for Primary Health Care Providers
available at https://www./tb/publications/ltbi/treatment.htm. Accessed May 22, 2019
24. Esmail H, Lai RP, Lesosky M, et al. Characterization of progressive HIV-associated tuberculosis using 2-deoxy-2-[18F]fluoro-D-glucose positron emission and computed tomography. Nat Med. 2016;22:1090-1093.
25. Ghesani N, Patrawalla A, Lardizabal A, et al: Increased cellular activity in thoracic lymph nodes in early human latent tuberculosis infection. Am J Respir Crit Care Med 189:748-750, 2014
26. Cox HS, Morrow M, Deutschmann PW. Long term efficacy of DOTS regimens for tuberculosis: systematic review. Br. Med. J. 2008; 336: 484–487
27. Malherbe ST, Shenai S, Ronacher K, et al: Persisting PET-CT lesion activity and M. tuberculosis mRNA after pulmonary tuberculosis cure. Nat Med 2:1094-1100, 2016
28. Steinbrück P, Dănkovã D, Edwards LB, et al. Tuberculosis risk in persons with 'fibrotic' x-ray lesions. Bull Int Union Tuberc. 1972;47:135-59.
29. Jeong YJ, Paeng JC, Nam HY, et al: (18)F-FDG positron-emission tomography/computed tomography findings of radiographic lesions suggesting old healed tuberculosis. J Korean Med Sci 29:386-391, 2014
30. World Health Organization. Treatment of tuberculosis guidelines, Fourth Edition.
Available at: https://www./tb/publications/2010/9789241547833/en/
Accessed October 11, 2015.
31. Sathekge MM, Ankrah AO, Lawal I, et al. Monitoring Response to Therapy. Semin Nucl Med. 2018;48:166-181.
32. Coleman MT, Chen RY, Lee M, et al. PET/CT imaging reveals a therapeutic response to oxazolidinones in macaques and humans with tuberculosis. Sci Transl Med; 2014; 6:265ra167.
33. Chen RY, Dodd LE, Lee M, et al. PET/CT imaging correlates with treatment outcome in patients with multidrug-resistant tuberculosis. Sci Transl Med 2014; 265:265ra166.
34. Martinez V, Castilla-Lievre MA, Guillet-Caruba C, et al. (18)F-FDG PET/CT in tuberculosis: an early non-invasive marker of therapeutic response. Int J Tuberc Lung Dis. 2012;16:1180-5.
35. Lefebvre N, Argemi X, Meyer N, et al. Clinical usefulness of 18F-FDG PET/CT for initial staging and assessment of treatment efficacy in patients with lymph node tuberculosis. Nucl Med Biol. 2017;50:17-24
36. Wahl R L, Jacene H, Kasamon Y, et al. From RECIST to PERCIST: Evolving considerations for PET response criteria in solid tumors. J Nucl Med 2009; 50 (Suppl 1): S122–S150.
37. Lee HS, Oh JY, Lee JH, et al. Response of pulmonary tuberculomas to anti-tuberculous treatment. Eur Respir J 2004;23:452–5.
38. Park IN, Ryu JS, Shim TS: Evaluation of therapeutic response of tuberculoma using F-18 FDG positron emission tomography. Clin Nucl Med 33:1-3, 2008
39. French MA. HIV/AIDS: immune reconstitution inflammatory syndrome: a reappraisal. Clin Infect Dis. 2009;48:101–107
40. Sathekge M, Maes A, Kgomo M, et al. FDG uptake in lymph-nodes of HIV+ and tuberculosis patients: implications for cancer staging. Q J Nucl Med Mol Imaging. 2010;54:698-703.
部分内容发表于:王毅翔. 18F-FDG-PET/CT在肺结核诊断及疗效评估中的应用 [J]. 临床与病理杂志, 2019. doi : 10.3978/j.issn.2095-6959.2019.